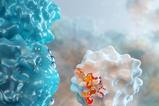
Scientists have developed a monoclonal antibody drug that could help to prevent infection from Acinetobacter baumannii.
University of Cambridge scientists have led the development of a monoclonal antibody drug that could help to prevent infection from Acinetobacter baumannii to combat the increasing and pertinent issue of microbial resistance.
A. baumannii bacteria, associated with hospital-acquired infections, can cause fatal respiratory illness and sepsis in vulnerable individuals, especially in newborn babies. Typically spread through contaminated surfaces, medical equipment and via contact with others, strains of this bacteria have become resistant to almost every available antibiotic.
Professor Stephen Baker from the Cambridge Institute of Therapeutic Immunology and Infectious Disease at the University of Cambridge commented: “The bacteria are naturally resistant to many antimicrobials, but as they’re now found in hospitals, they’ve acquired resistance to almost everything we can use. In some hospitals in Asia, where the infections are most common, there isn't a single antibiotic that will work against them. They’ve become impossible to treat.”
Transgenic mice
By using transgenic mice, the team demonstrated that the monoclonal antibodies could prevent infection with A. baumannii derived from clinical samples. First, the transgenic mice were exposed to the outer membrane of A. baumannii bacteria, which initiated an immune response. Following this, the scientists isolated almost 300 different antibodies and tested which of these was the most effective at recognising live bacteria. The single monoclonal antibody mAb1416 was identified as the best.
Mice were then treated with h mAb1416 and exposed them to A. baumannii, isolated from a child with sepsis in an intensive care unit, 24 hours later. They observed that the treated mice had a significant reduction in bacterial load in their lungs a further 24 hours later, compared to untreated mice.
Notably, the isolate used to test mAb1416 was taken from a patient ten years later than the other isolates which were from patients in Ho Chi Minh City, Vietnam. This demonstrates that mAb1416 was protective against A. baumannii bacteria that may have evolved over time.
mAb1416
Moving forward, further studies are required to elucidate the mechanism by which mAb1416 protects against infection, as this could enable the development of an even more effective treatment.
Professor Baker concluded: “We know that monoclonal antibodies are safe and that they work, and the technology exists to produce them – what we have done is identify how to hit bacteria with them. Apart from the cost effectiveness, there's no reason why this couldn’t become a medicine within a few years. Given the emergency presented by antimicrobial resistance, this could become a powerful new weapon to fight back.”
The research was funded by the Bill & Melinda Gates Foundation, the UK Medical Research Council Newton Fund, the Vietnam Ministry of Science and Technology, and Wellcome.
This study was published in Nature Communications.